What will happen if I agree to donate
If you do decide to donate, you will be given an information sheet to keep and be asked to sign a consent form. You will be asked about your lifestyle, past medical history and management of medical conditions. Your medical records will be examined and relevant data regarding your health and management of any medical conditions will be extracted and kept for reference in the future. The PCRFTB staff will also continue to monitor your future condition by enquiring through your medical records in hospital, with your General Practitioner (GP, Doctor) and also with Public Health England. This information will be anonymised so that you are not identified by other researchers. You will undergo the same treatment as a similar patient who is not taking part in the research, and you will be followed up in the clinic in exactly the same manner.
Whilst this process is underway we will request that you provide samples of blood (about four tablespoons) and urine (approximately 150 mL or a quarter pint) and saliva (spit). These samples will be collected at the same time as for your other tests.
If you subsequently undergo an operation on the pancreas, small volumes of blood, pancreatic juice and urine will be collected while you are asleep. While the operative procedure will not be modified in any way, small pieces of pancreatic and other adjoining organ tissues will be collected from the part of the organ that was already removed from you during the operation. The usual procedure is to send the excised tissues/organs for histological assessment (a range of tests to determine characteristics of disease under microscope) during the operation. This assessment will not be hampered in any manner. Usually, there is still some tissue remaining after these assessments. These tissues would be preserved in the PCRFTB and used for either immediate or future research. The range of use will be determined by the independent Tissue Access Committee.
During your regular follow-up visits, you will be invited to donate saliva, blood and urine specimens again.
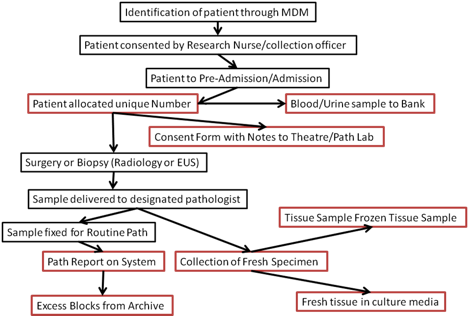
Pathways are designed, approved by Hampshire B Research Ethics Committee in 2014, so that participants can donate samples without compromising their personal data or interrupting their medical care. An example of this is summarised in the following flow chart.